We know patients with reduced kidney function need extra attention since they're at a higher risk of over-dosing, but how do we know if we've got it right?
Our simulations suggest yes, and model-informed dosing may offer a better path forward, even without therapeutic drug monitoring.
Here's what we found:
- Patients with reduced renal function are consistently overdosed with standard nomograms
- A simple model-based initial dose works well for patients with normal renal function
- Using model-informed precision dosing (MIPD) reduced neurotoxicity risk: for every 25 patients treated with MIPD, we could prevent one neurotoxicity event even approaching the likelihood neurotoxicity more conservatively than found in published literature
- MIPD combined with therapeutic drug monitoring provides the best balance of efficacy and safety, especially in patients with reduced renal function
The Cefepime Dosing Dilemma
Cefepime is widely used for treating severe infections caused by Pseudomonas aeruginosa and other gram-negative bacteria. Its broad-spectrum coverage makes it a go-to choice in critical situations. But choosing the right dose for each patient?
That's where things get tricky.
The challenge is balancing two competing priorities: hitting an efficacy target (keeping drug levels above the bacteria's minimum inhibitory concentration, or MIC, for as much of the dosing interval as possible), while also avoiding neurotoxicity from drug accumulation. This balancing act gets especially difficult when kidney function is reduced or fluctuating, which is common in critically ill patients.
Standard dosing nomograms (see examples from Northwestern and Stanford) adjust intervals based on creatinine clearance, but creatinine clearance doesn’t explain all the variability between patients. While extended infusions (3-4 hours) can help maximize the time above MIC, it may not fully correct the target attainment and toxicity balancing act in all patients. Therapeutic drug monitoring (TDM) paired with MIPD offers another strategy to personalize doses, but it's not yet clear which populations or clinical scenarios would benefit most from MIPD.
What We Tested
We simulated dosing for thousands of virtual patients with varying kidney function using established pharmacokinetic models by An et al. and Jonckheere et al. and compared three approaches:
- Standard Nomogram: Fixed doses with interval adjustments based on renal function
- Model-Based Initial Dose: PK model determines first dose, no subsequent adjustments
- MIPD with TDM: PK model determines first dose and subsequent MAP Bayesian estimation for dose adjustments following TDM after doses 2 and 3 for individualized adjustments
We tested these approaches across different renal function groups and infusion durations (short vs. extended). Our efficacy target was maintaining cefepime levels above either 1x or 1.5x an MIC of 8 mg/L, the current CLSI breakpoint for Pseudomonas. We evaluated target attainment after dose 5 and estimated neurotoxicity risk based on trough concentrations.
What We Found: Standard Dosing Overshoots in Reduced Kidney Function
A few patterns consistently emerged from our simulations:

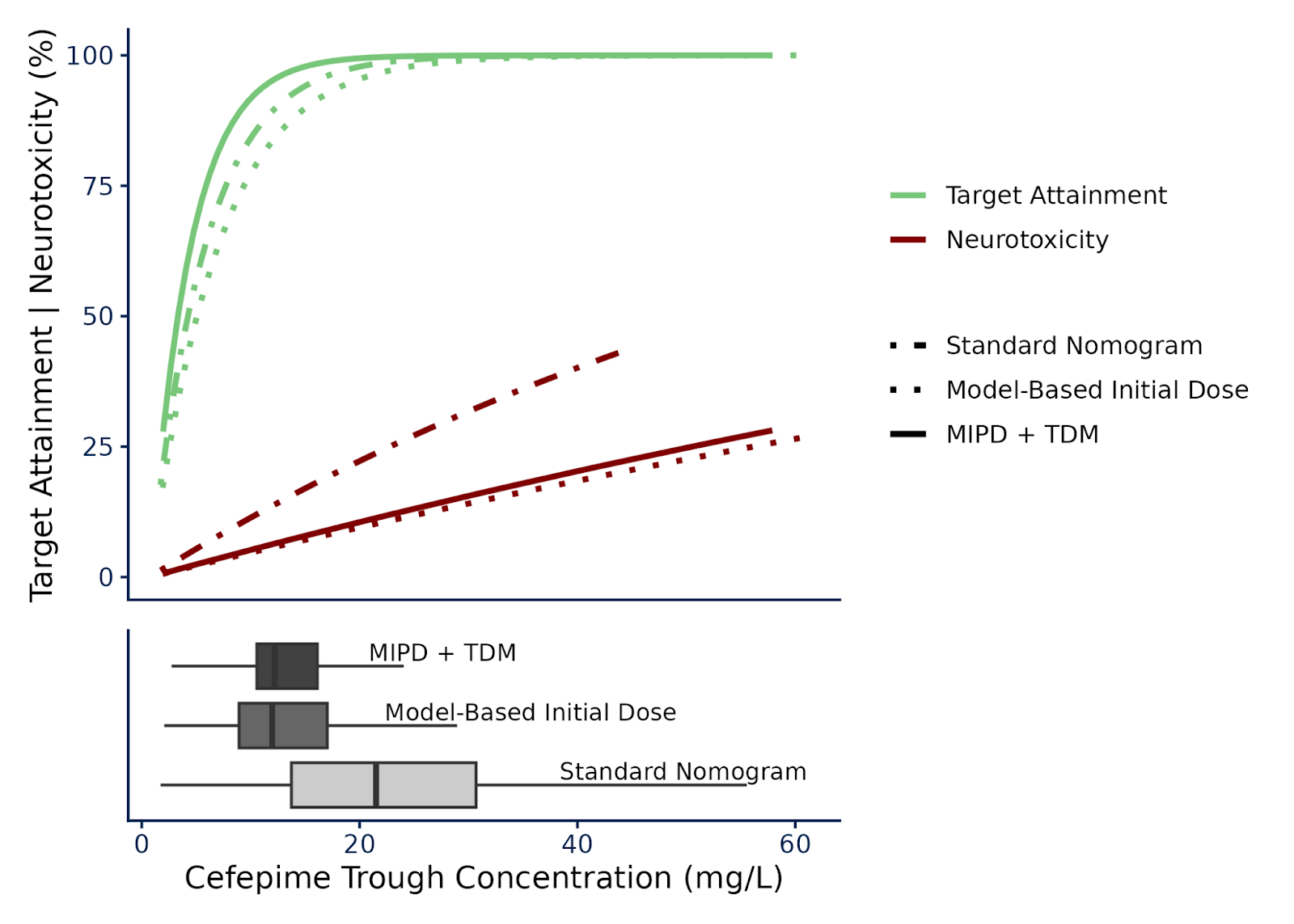
Figure 1. Cefepime steady state trough concentrations at steady state by dosing approach, infusion type, renal function, and target.
1. Standard Nomogram: High Doses, High Troughs, Mixed Results
Standard nomogram dosing achieved reasonable target attainment overall (an average of 81% of patients stayed above the MIC target), but the story varies dramatically by kidney function. Patients with creatinine clearance 30-60 mL/min showed excellent target attainment (98% average), but at a cost. These patients had notably higher doses and much higher steady-state troughs compared to MIPD approaches (Figure 1), raising concerns about unnecessary neurotoxicity risk.
On the flip side, patients with normal kidney function (>60 mL/min) receiving short infusions had poor target attainment (64% average), suggesting underdosing in this group.
The pattern is clear: interval-based dose reductions for patients with reduced kidney function often result in overexposure, while patients with normal function may be underdosed.
2. Model-Based Initial Dose: Decent in Normal Kidney Function
Using a PK model to calculate the first dose without subsequent TDM showed promise, but with caveats. When creatinine clearance is between 60 and 120 mL/min, a little over four out of every five patients stayed above the target concentration for 95% of the interval when targeting 1.5× MIC. When creatine clearance is above 120 mL/min or below 60 ml/min, this dipped to around three out of four patients.
In short, the current models tend to underestimate required doses in patients with reduced and elevated kidney function when you don't have actual concentration measurements to refine the predictions to the individual.
Why does model-based dosing sometimes miss the target for patients with poor renal function? We think it is because these patients are under-represented in the general patient population, and so models have fewer examples to “learn” from. As we have published previously, patient subgroup representation matters for model performance within that subgroup. Additionally, estimating how renal function relates with cefepime clearance using a more robust method such as CKD-EPI estimate of eGFR, as opposed to Cockcroft-Gault, may further assist in better accounting of the relationship.
So for these patients, model-based initial dosing results in a higher rate of under exposure but avoids those high troughs associated with neurotoxicity, while following the nomogram meant most patients hit the minimum target, at the cost of higher chances of neurotoxicity. How do we find the “Goldilocks” dose in the middle?
3. MIPD with TDM: Best Target Attainment and Safety Profile
When targeting 1.5× MIC (12 mg/L for an MIC of 8 mg/L), MIPD paired with TDM for Bayesian forecasting and dose adjustments achieved the target in an average of 94% of patients across all renal function groups and infusion types. Importantly, this approach delivered high target attainment while minimizing trough exposure, suggesting that improved efficacy does not need to come at the expense of safety. In fact, this approach consistently produced the lowest steady-state troughs compared with standard dosing, particularly in patients with creatinine clearance below 60 mL/min.
The neurotoxicity benefit was substantial: even when we assumed that we'd still need to treat fewer than 25 patients with MIPD instead of standard dosing to prevent one case of cefepime-induced neurotoxicity (Figure 2). More on this and how we accounted for the difficulties in predicting neurotoxicity shared below.

Figure 2. Pharmacodynamic endpoints of target attainment (fT>MIC > 95%) and neurotoxicity risk as approximated by a 0.50 multiplier (likely underpredicting) on the Boschung-Pasquier model over cefepime trough concentration and grouped by dosing approach when targeting 1.5xMIC. The fit curves are structured Percent = 100 × (1−e−k⋅Cmin,ss) with k estimated by endpoint and dosing approach with an asymptote at 100%.
Understanding Cefepime-Induced Neurotoxicity Risk
Cefepime-induced neurotoxicity (CIN) - including encephalopathy and seizures - is a major concern, and figuring out exactly how cefepime levels relate to neurotoxicity risk is still tough. One study by Boschung-Pasquier et al. found neurotoxicity beginning to appear at troughs as low as 7.7 mg/L, with all patients experiencing neurotoxicity when troughs exceeded 38.1 mg/L. This creates a troubling situation: clinical practice often aims to maximize time above 8 mg/L (or even 32 mg/L when targeting 4× MIC).
However, that study likely overestimated real-world neurotoxicity rates, a limitation the authors themselves acknowledged.
To account for this uncertainty, we calculated neurotoxicity risk using both the published estimates and a 50% reduction to represent a likely-underpredicting scenario. Even with the reduced risk estimate, moving from standard nomogram to either MIPD approach resulted in a Number Needed to Treat (NNT) of less than 25 patients to prevent one case of neurotoxicity across all combinations of renal function and infusion strategies.
What This Means for Practice
These simulations point to an important role for model-informed precision dosing of cefepime, particularly in two scenarios:
- Patients with reduced or fluctuating kidney function: MIPD with TDM provides the best combination of efficacy and safety. This is where standard nomograms are most likely to overshoot.
- Patients with stable normal kidney function: Model-based initial dosing (without subsequent TDM) is often a better option than standard nomograms to avoid underexposure (especially if receiving short infusions), though MIPD with TDM remains superior.
However, there are a few important caveats:
Our simulations assumed kidney function stays constant, which rarely happens in real ICU patients. Dynamic renal function means patients might require more monitoring and dose adjustment, not less.
In our simulations, we only adjusted doses, not dosing intervals. In practice, platforms like InsightRX Nova allow adjustment of both dose quantity and dosing interval. This flexibility can help in finding the right dosing regimen for your patient.
Clinical studies are still necessary to better understand the optimal exposure targets for cefepime. Targeting 1.5× MIC (e.g., 12 mg/L for an MIC of 8 mg/L) provided the best balance in our simulations, after accounting for patient variability; aiming directly at 8 mg/L lead to a greater percentage of patients undershooting the target. Targeting 4× MIC approximately doubled doses relative to standard nomograms, which seemed clinically unreasonable. As MIPD for cefepime scales up, it’s expected that we'll learn more about the right targets.
The Bottom Line
The optimal strategy we identified was MIPD plus TDM: 95% of individuals achieved therapeutic targets with a NNT of less than 25 to prevent one neurotoxicity event.
For cefepime, one size doesn't fit all. Standard dosing nomograms systematically overdose patients with reduced kidney function while sometimes underdosing those with normal function.
Cefepime MIPD is still in its early stages compared to more established applications like vancomycin; these simulations provide evidence that it can be a powerful tool to support dosing decisions for dynamic patients.